Stingray Behavior and Biology
Who are the Stingrays?
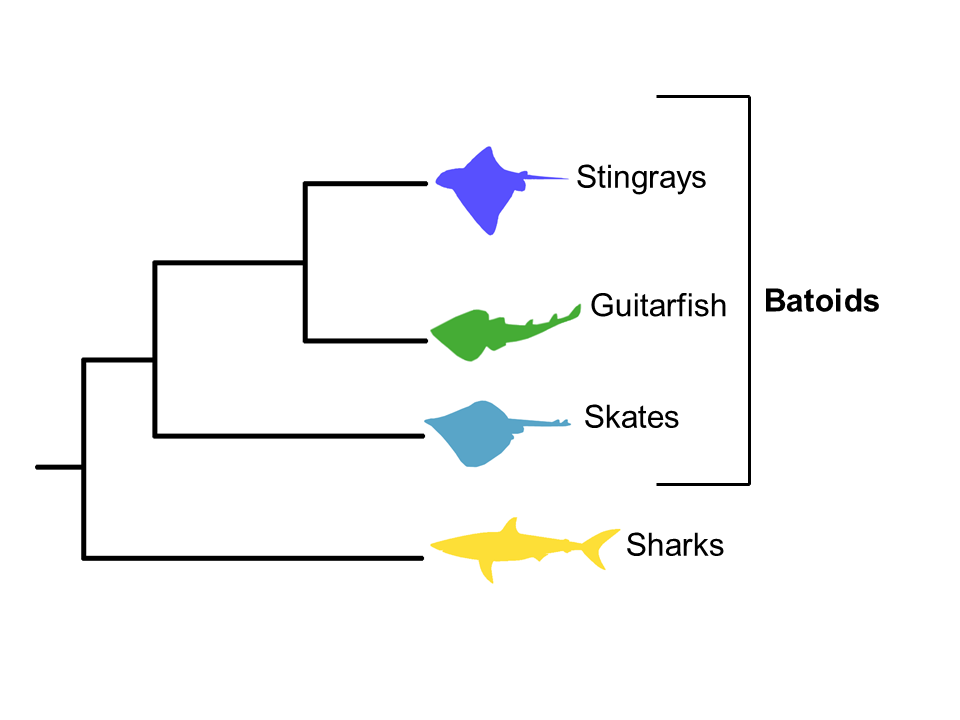
The stingrays are part of a unique group of fishes known as “batoids” and are closely related to sharks.
A stingray’s body is made of cartilage like a shark’s body so sometimes they are called “flat sharks”!
For more information check out the Chondrichthyan Tree of Life.
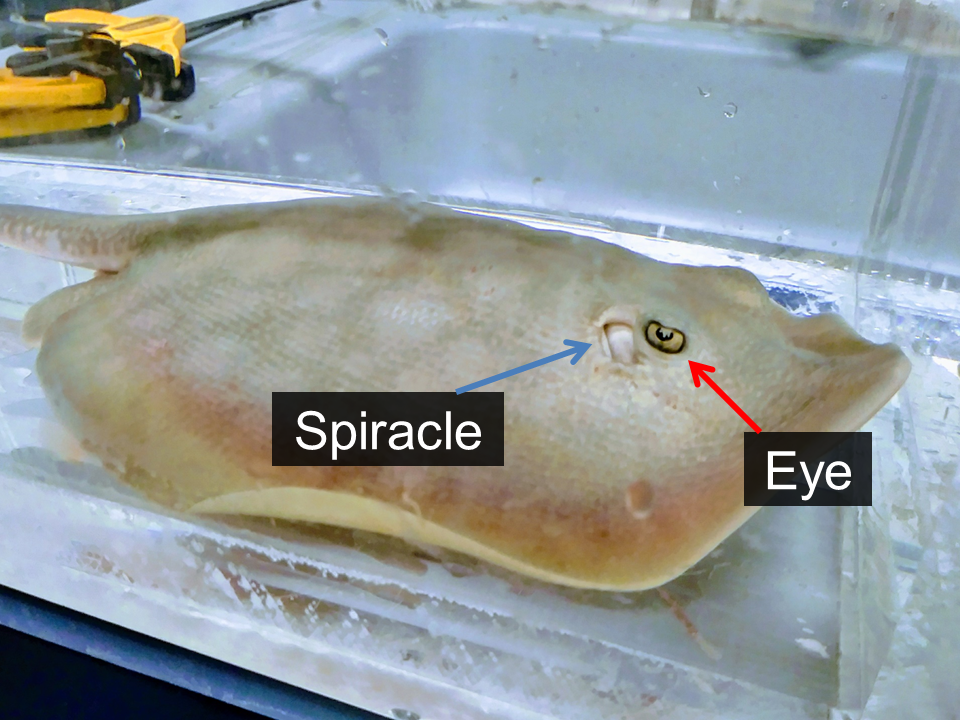
Most batoids spend their time resting on the seafloor with their mouths and gills on the bottom and their eyes and spiracles (snorkel opening for breathing) on top of their heads. Spiracles allow them to draw in clean water across their gills so they can breath while laying on the bottom.
Most stingrays forage on the seafloor for clams, marine worms, shrimp, isopods, crabs, and small fish.

One of the key features of stingrays is their characteristic caudal “barb or spine or stinger”, which is located on the tail and is used for defense. The barb is a modified dermal denticle which is seasonally shed in many species.

A stingray’s barb is covered by a thin external layer of skin and mucus. There are specialized cells in this outer layer of skin that allow for the transfer of venom into a potential predator upon striking.

A sting from a stingray can cause intense pain and throbbing – even the toughest surfer dudes have been brought to tears by a stingray’s sting.
How to Avoid Being Stung by a Stingray
Many stingrays bury themselves in the sand to remain hidden from potential predators while they rest or digest their food.

Water visibility during the summer at most southern California beaches is low making it harder to spot stingrays hidden in the sand. Try finding a stingray when it looks like this!

To avoid getting accidentally stung by a stingray, shuffle your feet through the sand to alert the buried ray that you are approaching and to give them the chance to skitter away (Lowe et al. 2007)

Beachgoers should follow all posted warnings and information which is usually found at the entrances of beaches. A lifeguard can also provide more information about current conditions and wildlife to watch out for at their beach if you ask!

Most stingray injuries are reported during the afternoon.
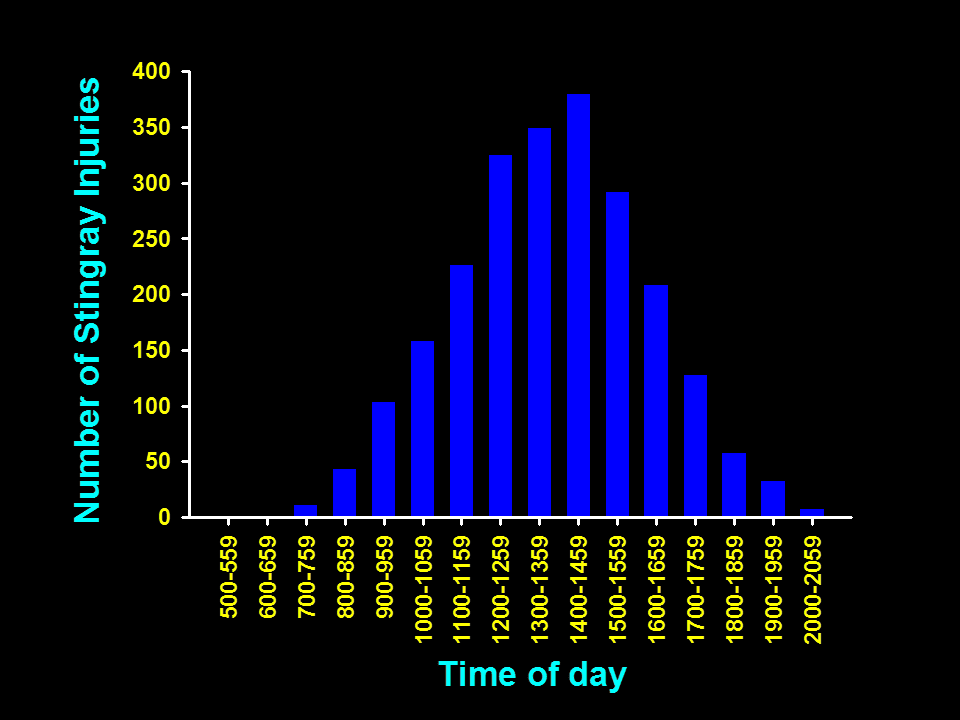
Most stingray related injuries occur in the afternoon, especially when the tide is lower and the water is warmer. This is likely because that is when there are the most people using the beach.
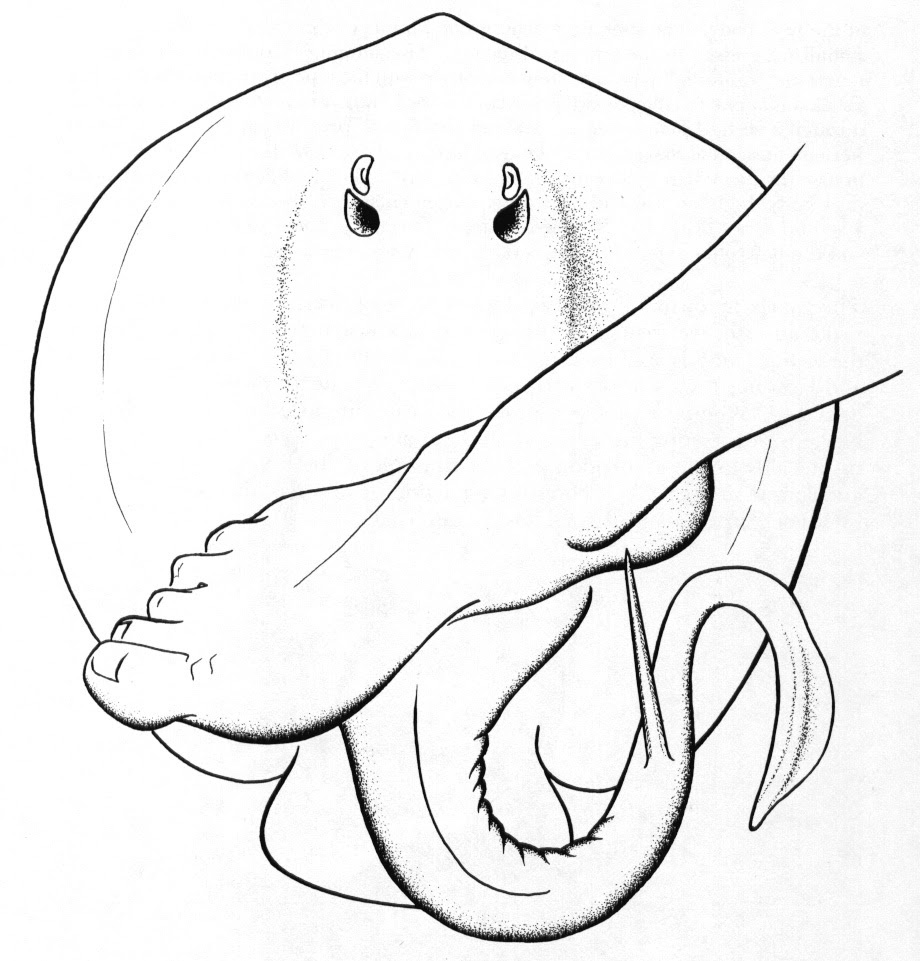
The most common place to be stung by a stingray is on your feet. When a stingray defends itself they whip their tail from side to side in an action called “spining”.
If you are stung by a stingray tell a lifeguard right away!

Hot water (not scalding) makes the pain from a stingray go away by breaking down the toxin. This may take more than an hour of soaking, so be patient. Also, remember that a stingray injury causes a laceration so make sure you disinfect it after soaking and keep an eye out for later infection.
Like what you see? Keep scrolling for more stingray research!
Round Stingray (Urobatis halleri)
The CSULB Shark Lab has conducted several studies on a particular species of ray known as the round stingray (Urobatis halleri), the most abundant ray species in Southern California!

| Kingdom | Animalia |
|---|---|
| Phylum | Chordata |
| Class | Chondrichthyes |
| Order | Myliobatiformes |
| Family | Urotrygonidae |
| Genus | Urobatis |
| Species | Halleri |
- Slow growing batoid species
- Reaches sexual maturity at about 4 years
- Live up to 14+ years
- Females give birth to litters of 1-6 pups and have one of the shortest gestation periods of any elasmobranch (only 3-4 months)
- Courting and mating occur in April-May, pupping occurs in Aug-Sep
- Males and females sexually segregate during non-mating season
- Diet consists of polychaete worms as juveniles and transitions to a mainly bivalve and crustacean diet as adults
Why study round stingrays?
- Highly abundant in Southern California
- Females have a short gestation period
- Easy to maintain in captivity
The round stingray is an ideal study species!

Human and Stingray Interactions
One of our goals in studying round stingrays is to share knowledge with the public in order to improve beach safety.
There is growing public concern as the number of stingray related injuries reported nationally each year has increased with thousands of cases being reported each year.

The historic depletion of large predators of round stingrays (sharks, giant sea bass, sea lions) has resulted in a population left largely unchecked resulting in population increases over the last 50 years. In addition, the loss of estuarine habitat used by round stingrays has driven populations to seek out suitable habitat along coastal beaches in closer proximity to people.
Due to the high population density of round stingrays and the increasing number of people visiting beaches each year, more interactions between stingrays and people are likely.

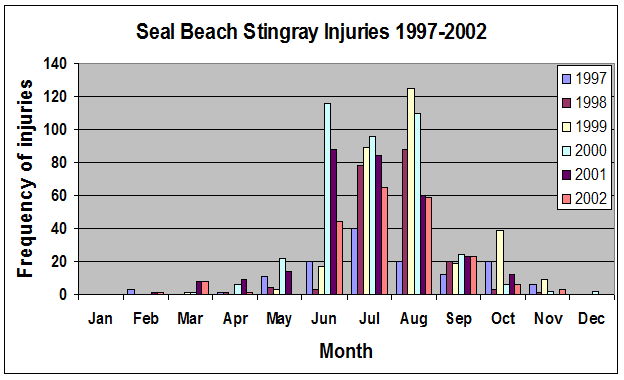
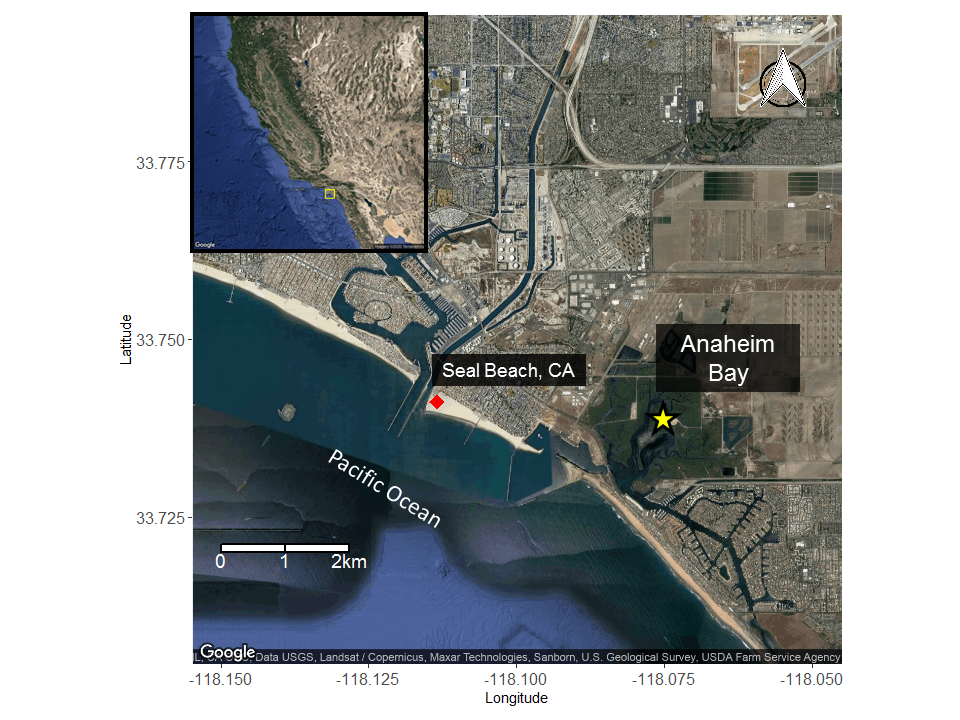
Many of our studies on round stingrays have been conducted in Seal Beach, CA where round stingrays are notoriously abundant, earning it the nickname “Ray Bay”.
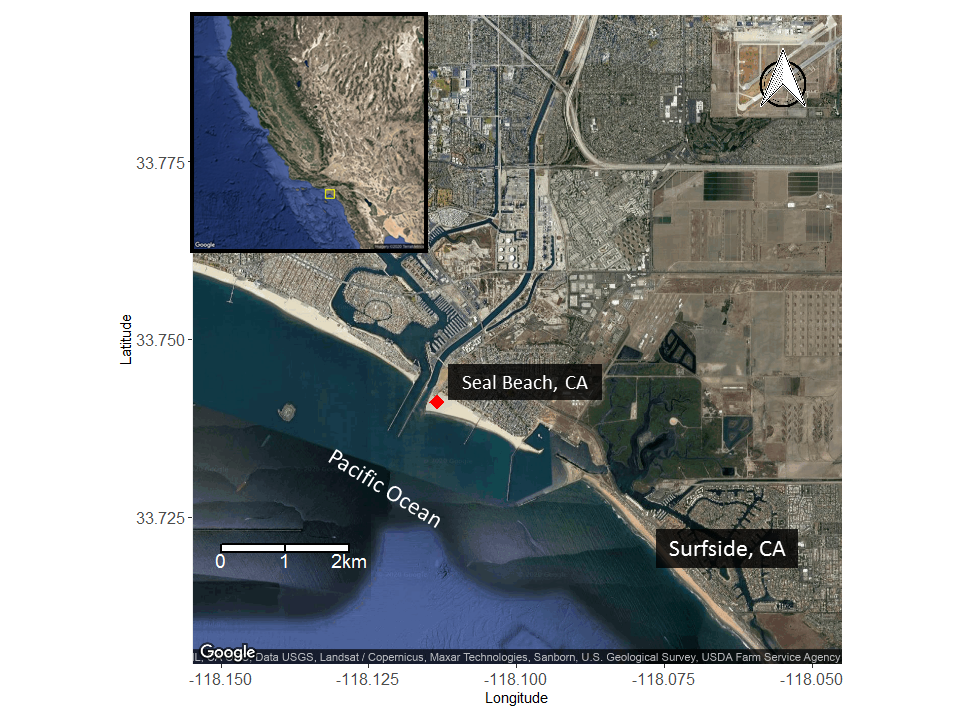
Beachgoers are attracted to Seal Beach for a variety of aquatic activities including surfing, wading, swimming, and bodysurfing.

There are thousands of visitors to southern California beaches each summer. Beaches are especially crowded on Labor Day weekend.

As more research on round stingrays is conducted we can make better informed decisions about how to share the marine environment.
Round Stingray Abundance and Distribution
Seal Beach, CA is located near the mouth of the San Gabriel River where two electrical generating stations release seawater that is used for cooling the stations. The seawater released from these stations after use has a higher temperature than what is naturally found in the river which then makes its way towards Seal Beach.
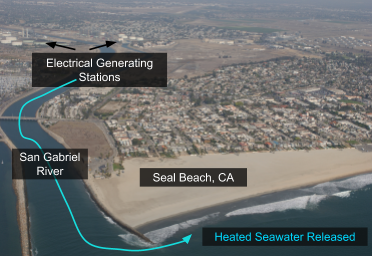
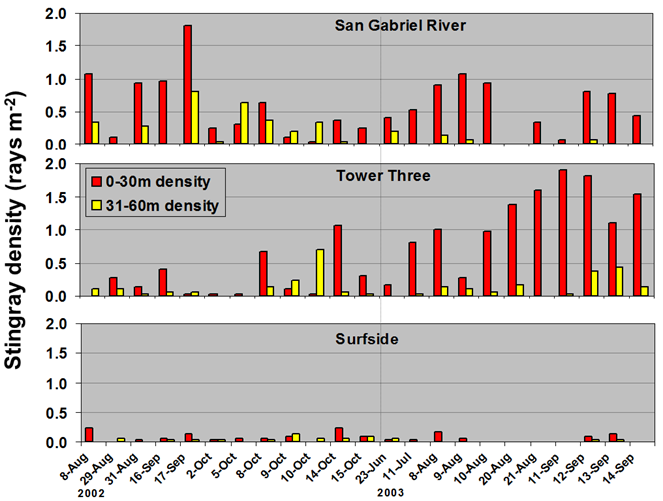
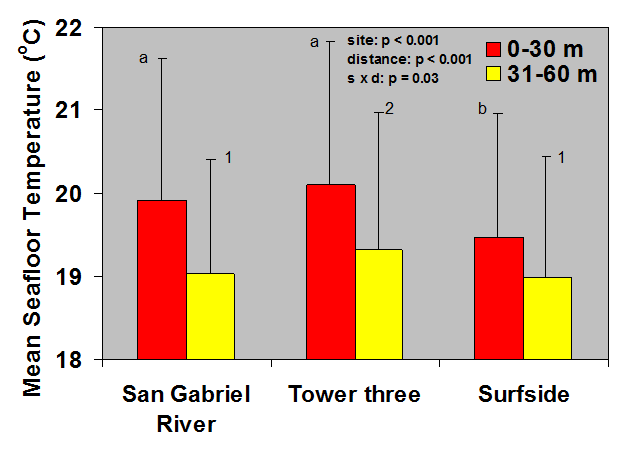
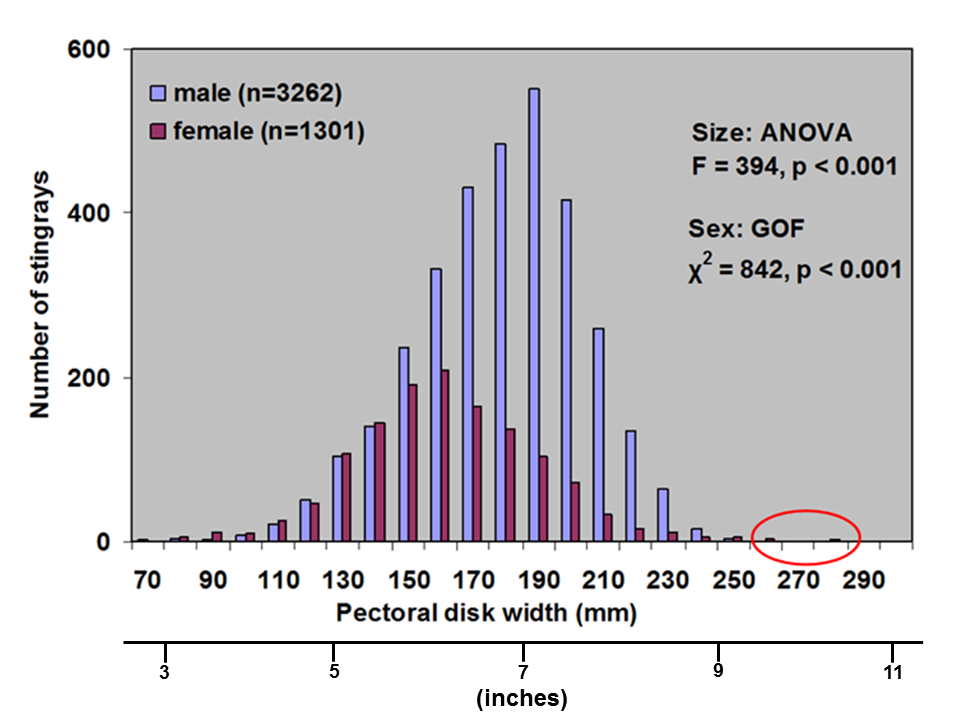
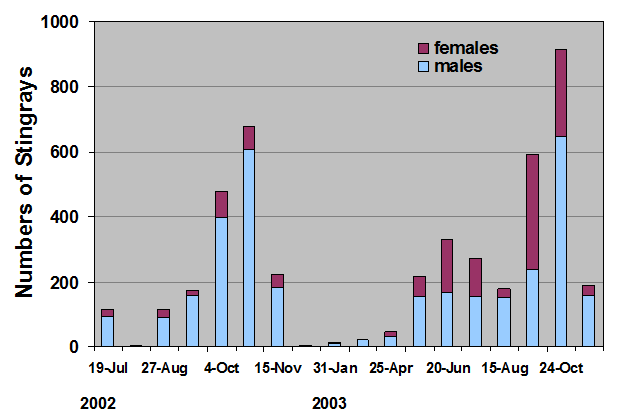
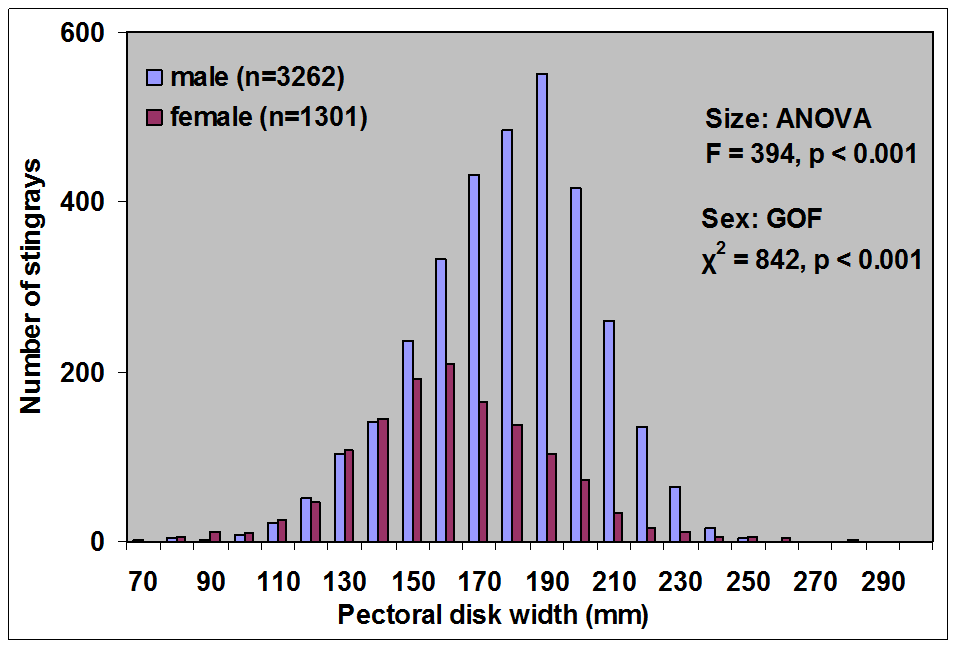
Because of this input of very warm water to the local environment more stingrays are attracted to the area. Hoisington & Lowe 2005 conducted a study to examine the abundance and distribution of round stingrays (U. halleri) in Seal Beach near this warm water outfall (Fig. 6).
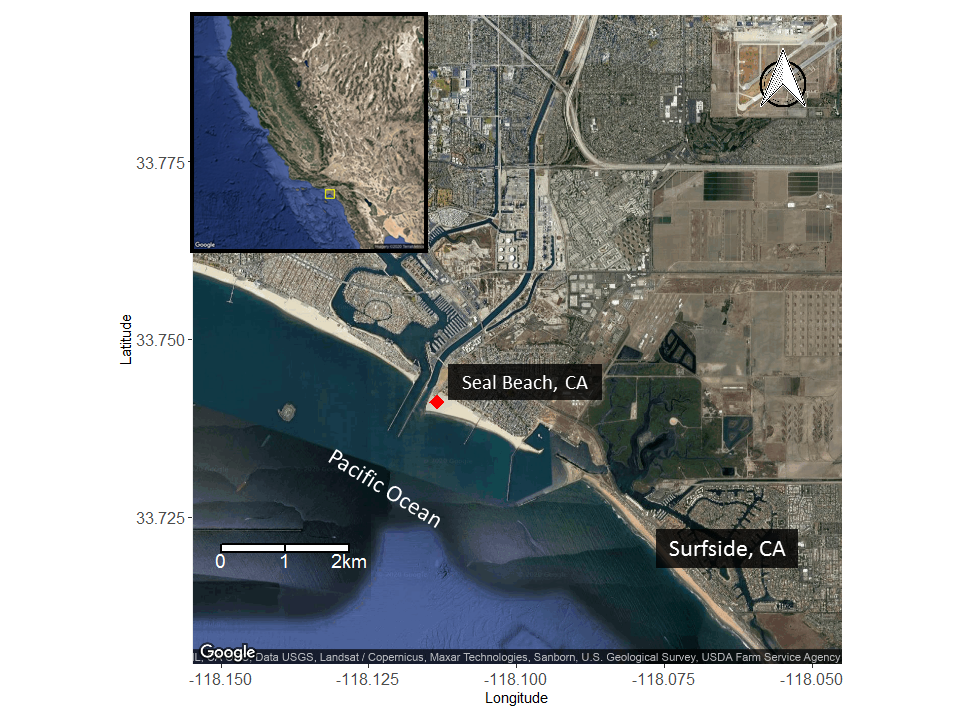
Hoisington & Lowe 2005 found that round stingray abundance is greatest within the first 30m (approx. 100 ft.) from shore where most beachgoers are found. Abundance was also greater in Seal Beach when compared to the nearby control site in Surfside, CA which does not receive an input of warm water.
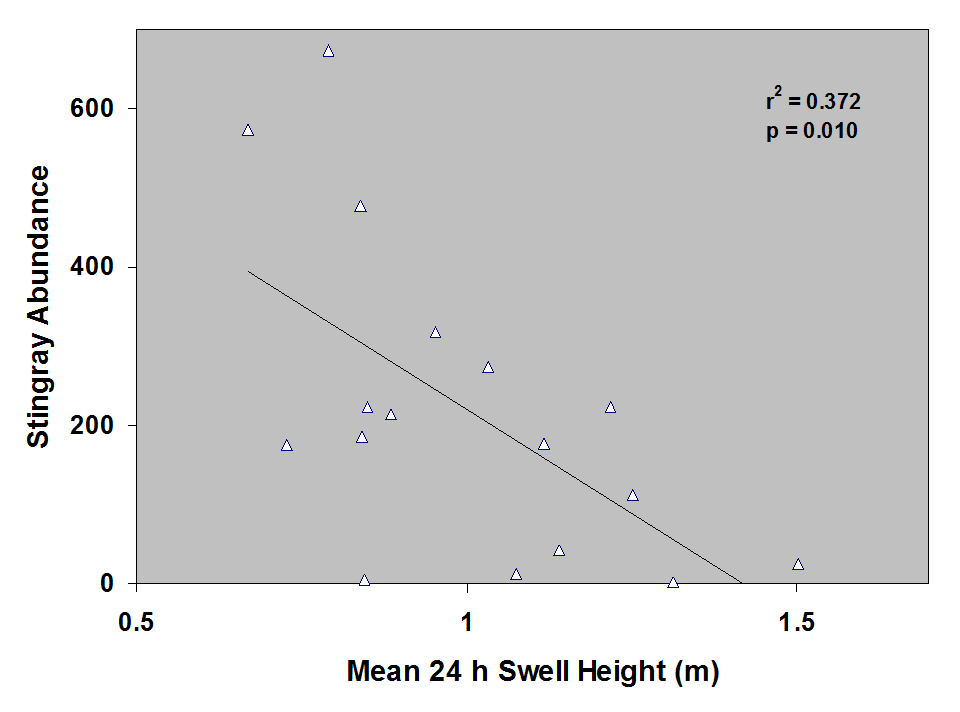
Round stingray abundance during sampling was negatively correlated with swell height (i.e. as swell height increases abundance decreases). Abundance within the first 30 m from shore is greater when conditions are calm (i.e. low swell, Fig. 14).
Hoisington & Lowe 2005 found that round stingray abundance is seasonal and increases in summer months with peak abundance in the fall (Fig. 15).
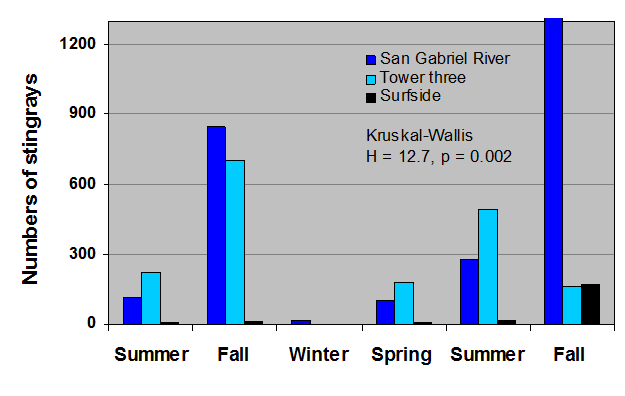
During most times of the year male round stingrays are more abundant nearshore, but between the months of June and September-October female stingray abundance increases. This is likely due to mating during the early summer and for a reproductive benefit to females in the fall.
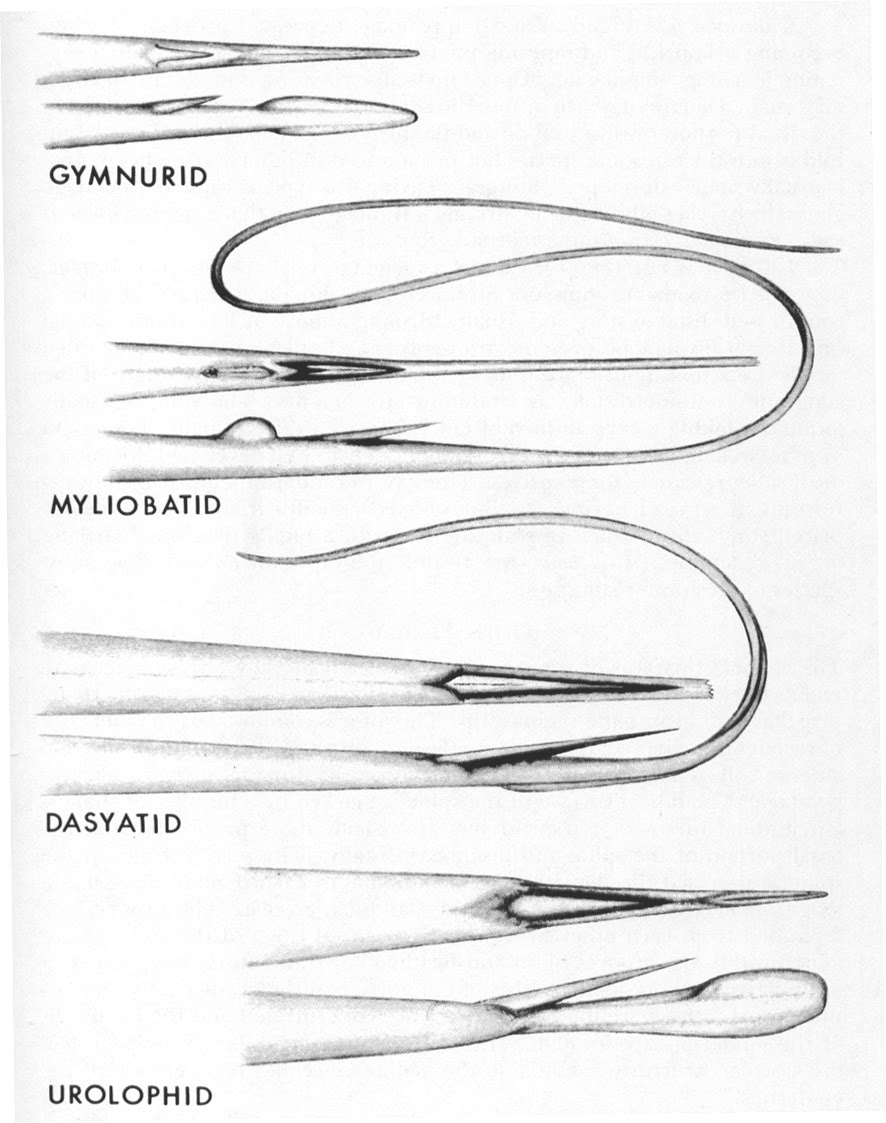
Round Stingray Spines
One of the key features of the batoids group (stingrays) is their characteristic “caudal spine”, sometimes referred to as a “barb”, which is located on the tail and is used for defense. The caudal “spine” is actually a modified placoid scale similar to the scales found on the skin of rays and other elasmobranchs.
The spine is a unique weapon in that it not only can puncture or lacerate its attacker, but also can transmit a venom from the mucous coating on the spine via grooves running along the ventral side. This venom can cause intense pain and throbbing – even the toughest surfers have been brought to tears by stingrays.

Stingray spines vary in length and position depending upon species.
Stingray spines are like fingernails, in that they lack nerves and grow back after a period of time (Lowe et al. 2007).


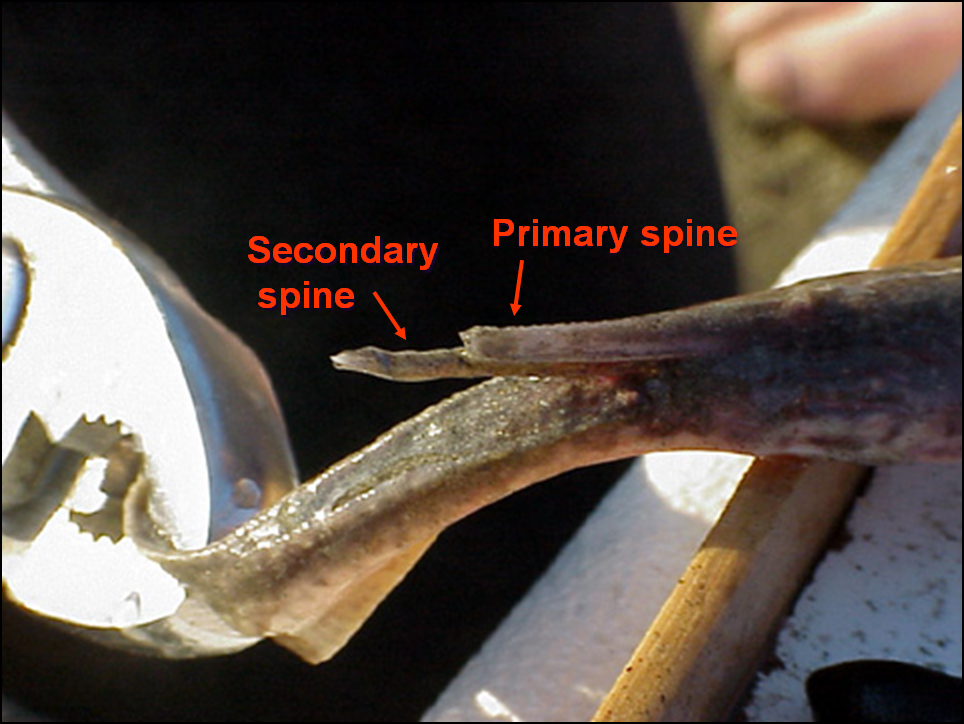

Round stingrays possess the ability to replace damaged or lost caudal spines outside of the typical shedding period.

Round Stingray Tooth Morphology
Undergraduate student Justin Hackett examined how tooth morphology varies across age and sex of round stingrays. Results showed that round stingray dentition is most influenced by age and sex.
Prior to maturity (DW < 15 cm), juvenile male and female round stingrays do not exhibit significant differences in tooth morphology as both sexes share similar diets and habitat.
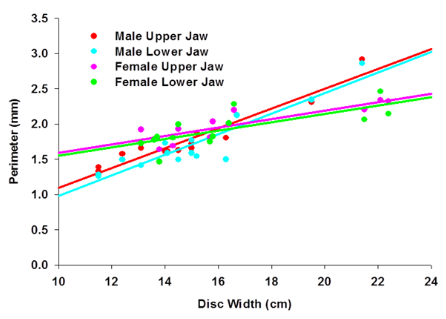
Female round stingray teeth are rounded and diamond shaped. Females do not exhibit differences in tooth shape between juveniles and adult stages, but mature females have significantly larger teeth than juvenile females.

Mature male round stingray teeth are larger than mature female round stingray teeth and become more triangular in shape only during the breeding season, as males use their teeth to hold females for during copulation. After mating season, male round stingray detention returns to flat, plate-like teeth used for crushing clams and crustaceans.

Round Stingray Spine Clipping Study
The city of Seal Beach, CA has historically implemented various strategies in order to help reduce the number of stingray related injuries. Many proposed programs did not have a significant impact on reducing the number of stingray related injuries and have been discontinued.
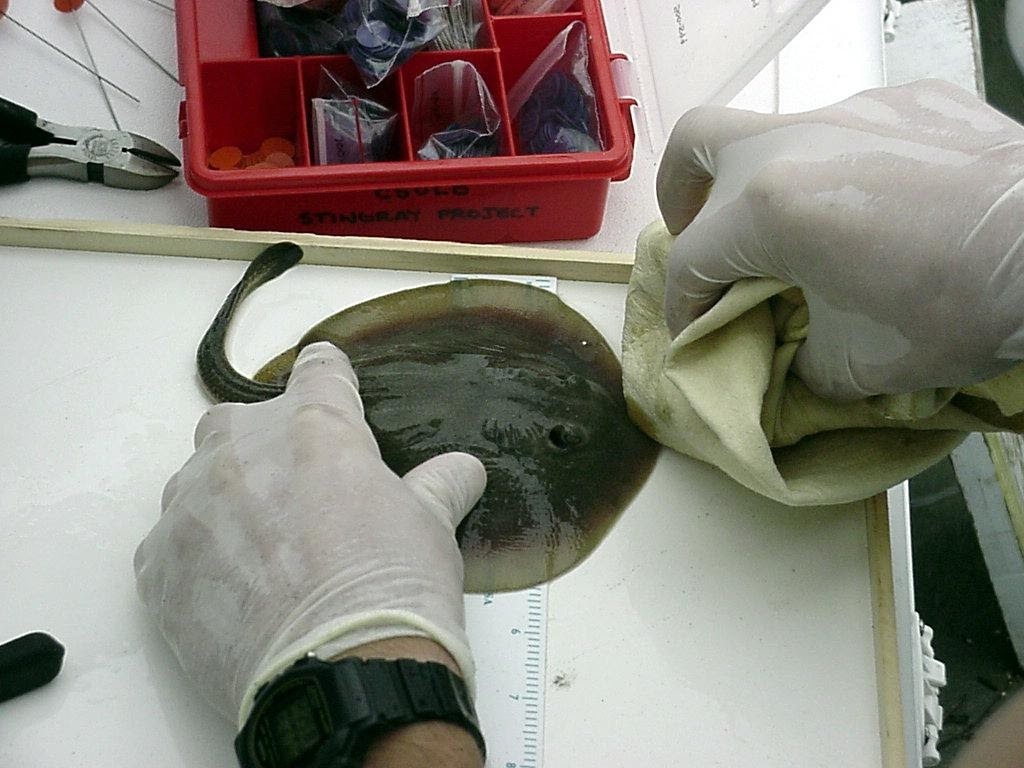
Lowe et al. 2007 conducted a study to investigate the effectiveness of a stingray spine clipping program in reducing the number of stingray related injuries.



| Date | Stingrays Tagged | Stingrays Recaptured |
|---|---|---|
| 06/08/2001 | 202 | |
| 08/03/2001 | 157 | 2 |
| Population Estimate (Lincoln-Petersen Method) |
~16,000 stingrays |
Follow up mark and recapture studies at Seal Beach reported low recapture rates despite a high sampling effort, indicating that the round stingray population in Seal Beach is very mobile with a high turnover rate (Vaudo and Lowe 2006, Lowe et al. 2007).
Given the size of the population and the amount of new stingrays that can be found in the area, particularly during the summer/fall months when stingray abundance is at its peak, a stingray spine clipping program was deemed impractical and unrealistic to implement.
Therefore, the best defense against stingray related injuries is to remember to shuffle your feet and to educate beach visitors about the local wildlife they may come into close contact with during their visit.

Round Stingray Movement and Site Fidelity
Vaudo and Lowe 2006 investigated the fine-scale movement patterns of round stingrays caught in Seal Beach near the mouth of the San Gabriel River and determined whether round stingrays exhibit any site fidelity to Seal Beach.

3 acoustic receivers were deployed in Seal Beach and 2 additional receivers deployed at neighboring sites Belmont Shore and Surfside.

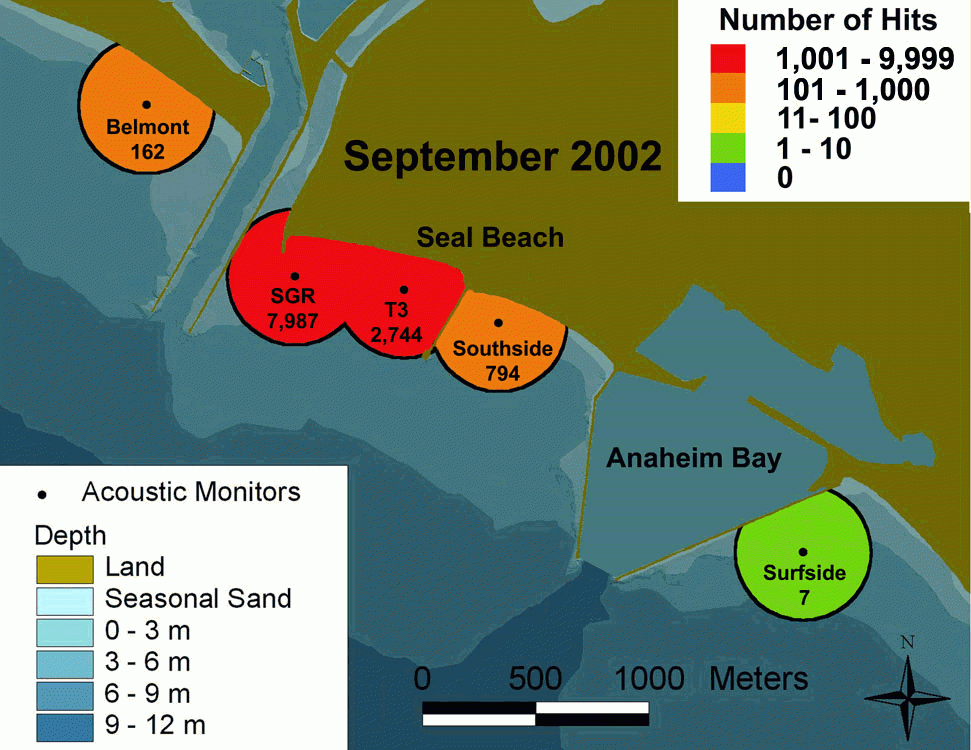
Round stingrays exhibit the potential to travel longer distances as was the case when one female tagged at Seal Beach was captured and released >30 km (15 mi) south in Upper Newport Bay two months later. The same female was then detected back at Seal Beach one year after initial tagging.
Fine-scale (precise) movement data was collected via active tracking following the emitted signal from a tagged individual using a directional hydrophone.

Active tracking showed that round stingrays exhibited short movements interspersed by 2-4 hr. periods of inactivity and that round stingray movement was greatest at night during the ebbing (falling) tide when water temperature can increase as much as 10 degrees (C) in a relatively short period of time. Possible explanations of this increased activity could be to find more desirable conditions, increase foraging success, or find potential mates.
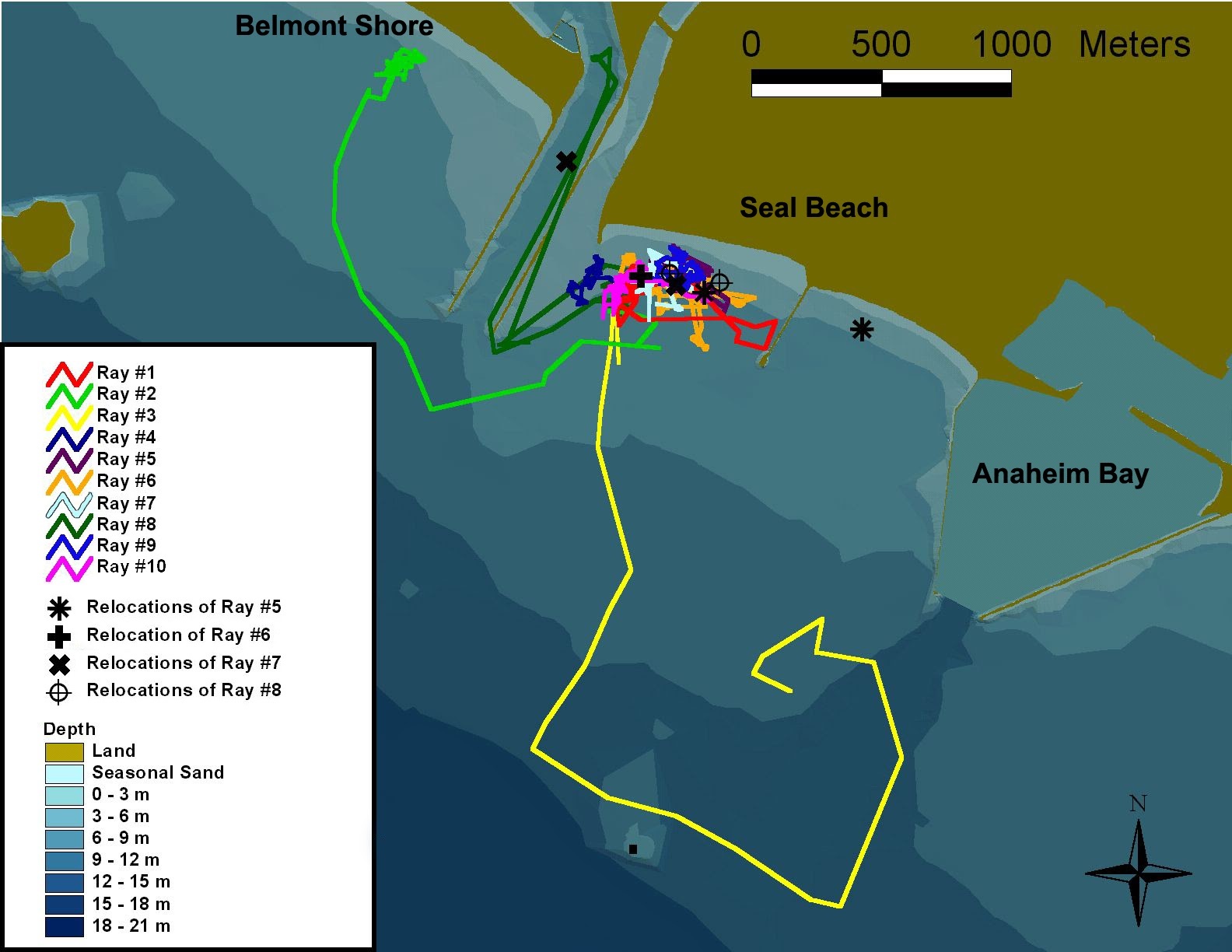
Vaudo and Lowe 2006 found that round stingrays seasonally congregate for a few weeks at Seal Beach before dispersing to other locations in search of suitable breeding grounds.
Round Stingray Age and Growth
Hale and Lowe 2008 conducted a growth and age structure study of the round stingray (U. halleri) population at Seal Beach, CA. Estimates of age were determined by analysing the vertebral growth rings of round stingrays (U. halleri) collected from Seal Beach. Growth rate coefficient (K) and theoretical maximum disc width (WD) were estimated using the von Bertalanffy growth model.
This information was used in conjunction with size-frequency data from periodic sampling conducted at Seal Beach to determine age structure of the local round stingray population. From this study it was found that the population of round stingrays at Seal Beach is composed mostly of sexually mature adults. Additional findings were that round stingrays are slow growing relative to other species in the family Urolophidae.
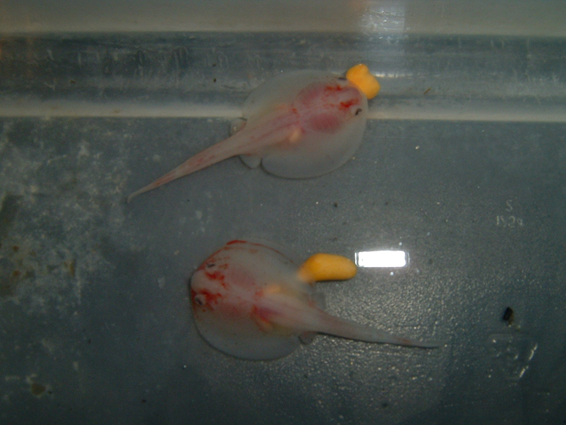
Round Stingray Reproduction
Life History
Round stingrays have an annual reproductive cycle with peaks in mating occurring from May-July. Round stingrays have a gestation period of 3-4 months and may give birth to litters of 1-6 pups. Birth occurs from August - November (Babel 1967, Mull et al. 2010(a), Lyons and Lowe 2013).


Round stingrays are aplacental viviparous (ovoviviparous), meaning they give birth to live young and embryos do not have an attached placenta (Lyons and Lowe 2013).
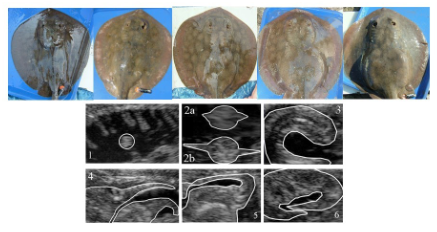
Jirik and Lowe (2012) used ultrasonography to examine the condition of pregnant females non-invasively.
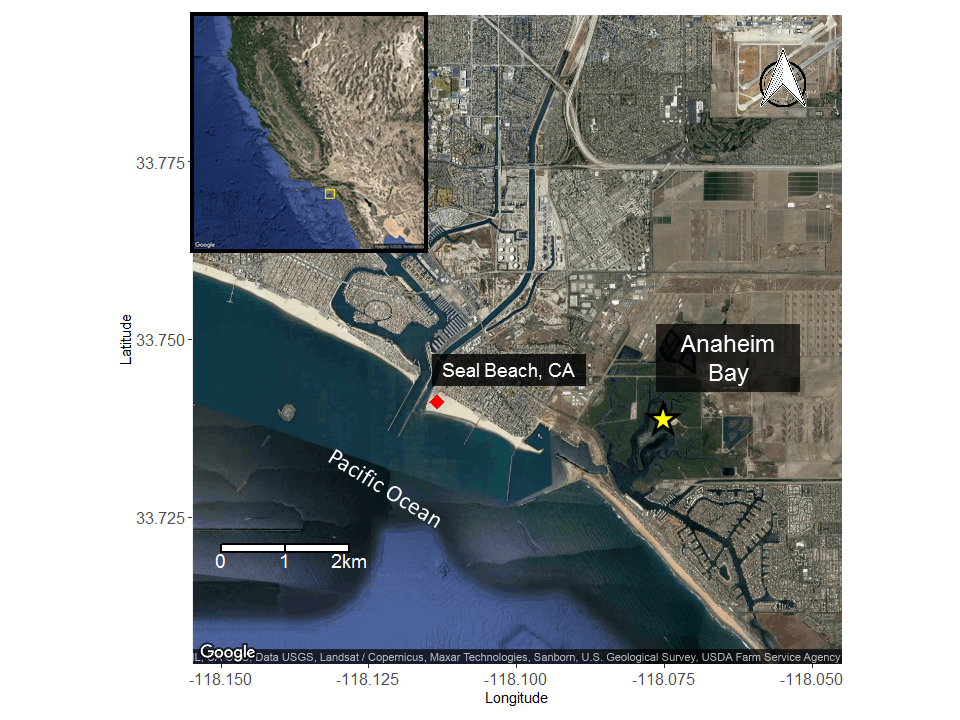
Seasonal Movement Patterns
Females sexually segregate to avoid aggressive interactions during non-mating seasons and to seek warmer coastal waters during gestation (Hoisington and Lowe 2005, Mull et al. 2010, Jirik and Lowe 2012). Males are not found in warmer coastal waters as much as females possibly due to a sex-specific energetic cost from residing in areas of increased temperatures for extended periods (Jirik and Lowe 2012).

Anaheim Bay is a restored estuarine habitat and is a proposed thermal refuge for female round stingrays (Jirik and Lowe 2012).
Multiple Paternity in Round Stingrays
Multiple paternity in elasmobranchs is relatively common and is due to competition that arises from sexual selection both among (intersexual) and within sexes (intrasexual).
Female round stingrays have two uterine chambers and can accommodate multiple fetuses at varying stages of development.
Lyons and Lowe 2017 found that 90% of round stingray litters examined contained multiple fetuses sired by multiple fathers.
Maternal Offloading of Organic Contaminants
Organic contaminants (OC) such as PCBs, DDT and its derivatives (DDE and DDD) are problematic in the marine environment. These compounds are resistant to biodegradation, lipophilic (fat soluble), and biomagnify in the fatty tissue of upper trophic level predators including elasmobranchs. In female elasmobranchs, contaminants are transferred to developing embryos through the mobilization of hepatic (liver) lipids to the embryo through a variety of nutrient supplementing pathways depending upon reproductive mode.
Lyons and Lowe 2013 used round stingrays as a model species to determine pathways of contaminant transfer, the quantity of contaminants offloaded by females, and the relative proportion of organic contaminant groups (PCBs, DDTs, and chlordanes) transferred from mother to embryo.

Lyons and Lowe 2013 found that female round stingrays transferred contaminants throughout pregnancy and that ova development and the delivery of histotrophic fluid to embryos were the two pathways that significantly contributed to offloaded contaminants in offspring. The mother’s total contaminant load as measured by the liver was found to be the most influential factor in predicting embryonic contaminant load while size/age did not strongly influence contaminant loads.

On average, 1.5 1.7 % (Mean ± SD) of total contaminants were offloaded from mother to embryo and contaminant loads were similar in concentration between embryos in litters. Mothers and embryos had similar contaminant profiles for the three contaminant groups studied with PCBs comprising >80% of contaminant load in embryos while chlordanes are the second largest contaminant group, and DDT was found in the lowest concentration.
Female round stingrays appear to offload less contaminants to their offspring via this mode of reproduction compared to other elasmobranch species. Further research should continue to examine maternal offloading in other elasmobranch reproductive modes and the physiological effects of contaminant exposure in developing embryos and neonates.
Male Reproductive Cycle
Mull et al. 2010(b) characterized the reproductive cycle of male round stingrays from Seal Beach, CA by examining changes in testes morphology, spermatogenesis, and steroid hormone concentrations throughout their reproductive cycle.
The reproductive cycle of male round stingrays is annual and many aspects of reproduction are highly regulated. Males experience increases in Gonadosomatic Index (GSI) from July-October with peak GSI in October. Testosterone (T) production remains low from April-June then increases until peak production in March. 11-Ketotestosterone (11-KT) concentrations followed a similar pattern but peak production occurred in February. Sperm production peaks in December and is stored long term until favorable mating conditions are present.
Additional findings were that GSI, and 11-KT are negatively correlated with increased day length but not temperature. However, T production is negatively correlated with both increased daylength and temperature evident from in-lab experiments where temperature was manipulated over a period of several weeks.

These findings support the idea that temperature plays an important role in the regulation of T and may serve as a cue for reproduction in male round stingrays.
Round Stingray Population Genetics
Microsatellite analysis of round stingrays collected from sites in Southern California, Gulf of California, and Santa Catalina Island to determine level connectivity and structure was conducted by Plank et al. 2010.
It was found that the Santa Catalina Island population of round stingrays was genetically distinct from populations of round stingrays found in Southern California and the Gulf of California.
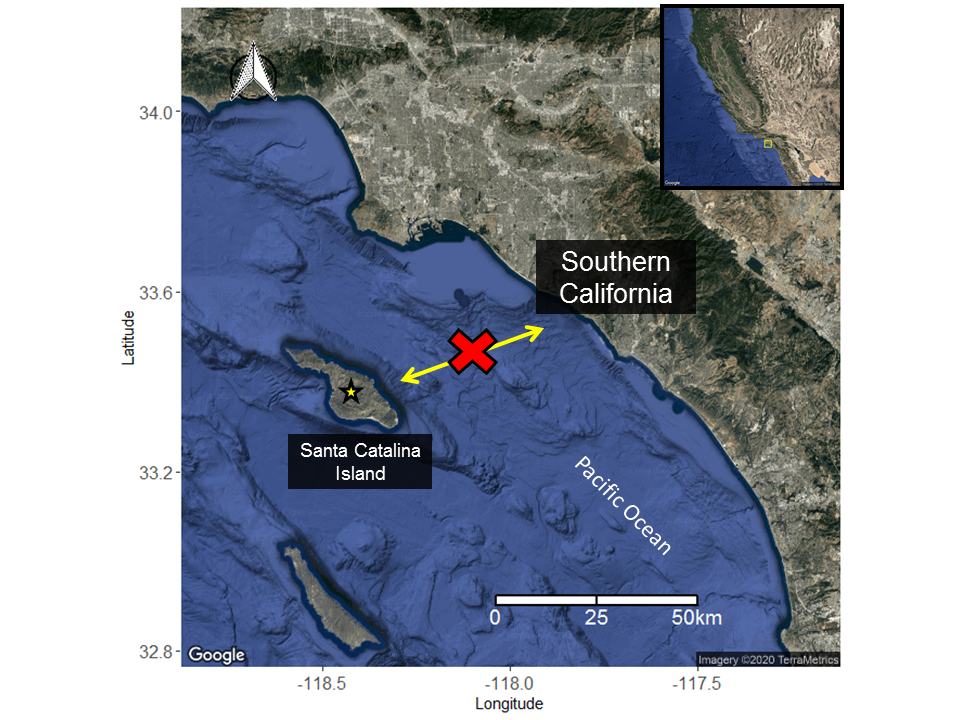
Santa Catalina Island round stingrays are genetically distinct from populations of mainland round stingrays and exhibited the lowest genetic diversity among the treatment groups suggesting a recent founder’s effect in the island population. The earliest known records of round stingrays on Santa Catalina Island is 1970.
Additional findings were that populations of round stingrays in the eastern central Gulf of California are not isolated from populations on the Pacific coast of Baja California.
Metabolic Rates of Round Stingrays
Graduate student Lorena Silva Garay is investigating the effects of size and temperature on the metabolic rate of round stingrays across its entire size range. The goals of her project are to estimate the Standard Metabolic Rate (SMR) and metabolic Q10 of round stingrays across a range of temperatures and predict the species-specific scaling exponent of metabolic rate for round stingrays. This is one of the first studies to estimate these parameters across the entire size range of an elasmobranch species.
Standard Metabolic Rate and Q10
Standard Metabolic Rate (SMR) of round stingrays was measured using the mass-specific oxygen consumption rate (MO2, mgO2 kg−1 h−1) from static respirometry trials. Trials were conducted at three temperatures spanning the seasonal temperature range experienced by round stingrays in southern California (15°, 23°, 27°C).



Silva found that SMR increased with increasing temperature more so in juveniles than in adults, and did not significantly vary between sexes. In adult individuals a plateauing of SMR with increasing temperature was observed.
While Q10 did not significantly vary between sexes, juvenile and adult round stingrays exhibited significant differences in thermal sensitivity; juveniles displayed a higher Q10 than was estimated for adults when accounting for the interacting effects of temperature and body mass.
The overall thermal sensitivity of round stingrays is relatively higher than previously reported values for other elasmobranchs. In adult round stingrays, the combined results of a plateauing SMR with increased temperature and relatively lower Q10 is likely indicative of a broader range of thermal optima for larger individuals. The broader thermal optima in adult round stingrays is consistent with the seasonal changes in water temperature experienced in their environment.
For juveniles, the higher SMR and Q10 indicates that juvenile round stingrays are more sensitive to shifts in temperature and exhibit a narrower range of thermal optima unlike adult round stingrays. Thus, it is likely that smaller juveniles would display a preference for cooler waters that maximizes food intake, assimilation, and growth against energetic cost. Given their high thermal sensitivity, juvenile round stingrays are predicted to be most impacted by increases in ocean temperatures.
Because sex was not a significant factor in predicting differences in thermal sensitivity and SMR of round stingrays, this indicates that the seasonal sexual segregation of mature females of the species in southern California is likely the result of a trade-off between mating opportunity and metabolic costs.
Contaminants in Round Stingrays
Lyons et al. 2014 conducted a study with the goals of comparing bioaccumulation patterns across age and sex, quantifying biochemical response to contaminant exposure, and comparing differences in accumulation patterns from stingray populations on nearby Santa Catalina Island and mainland southern California.
Differences in Contaminant Accumulation by Age and Sex
Results showed that juveniles did not show differences in accumulation between sexes. Juvenile males in particular showed a significant decrease in contaminant concentration as they increased in size until maturity. This pattern of bioaccumulation is likely due to juveniles growing at a faster rate than their rate of contaminant intake. Additionally, differences in diet and habitat use between the two life stages could further explain differences in contaminant concentration.
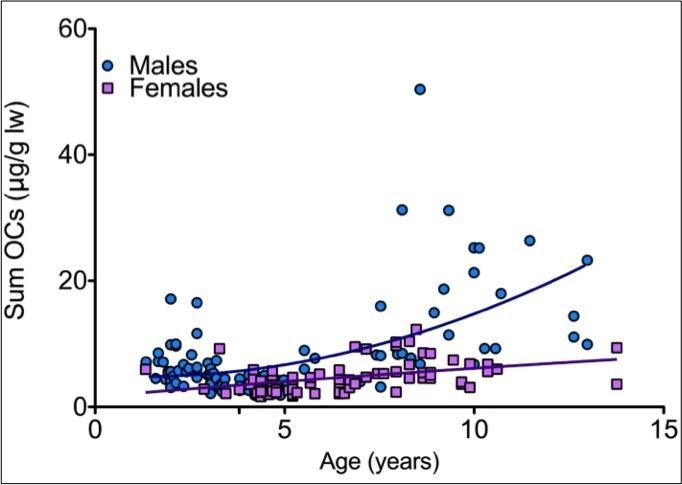
Mercury Accumulation in Round Stingrays
In a follow up study by Lyons et al. 2017, mercury accumulation in male round stingrays was examined to determine potential differences in bioaccumulation with ontogeny. It was found that liver concentrations of mercury dramatically increased after maturity and mature male round stingrays had significantly higher concentrations of mercury than juveniles. Differences in concentration are likely due to shifts in energetic costs as mature round stingrays partition more energy towards reproduction and have less energy to direct towards detoxification processes.
Biochemical Response
Lyons et al. 2014 examined bioaccumulation patterns across age and size of round stingrays and quantified biochemical response using ethoxyresorufin-O-deethylase (EROD) activity to determine if contaminants have any negative effect on round stingray biology.
CYP1A is a useful biomarker of exposure that can be used to demonstrate biochemical responses to certain classes of organic contaminants. The activation of this protein is often measured in terms of ethoxyresorufin-O-deethylase (EROD) activity. Quantifying CYP1A activity through EROD assay is a widely used technique to indirectly measure the amount of expressed enzyme and therefore an organism’s response.
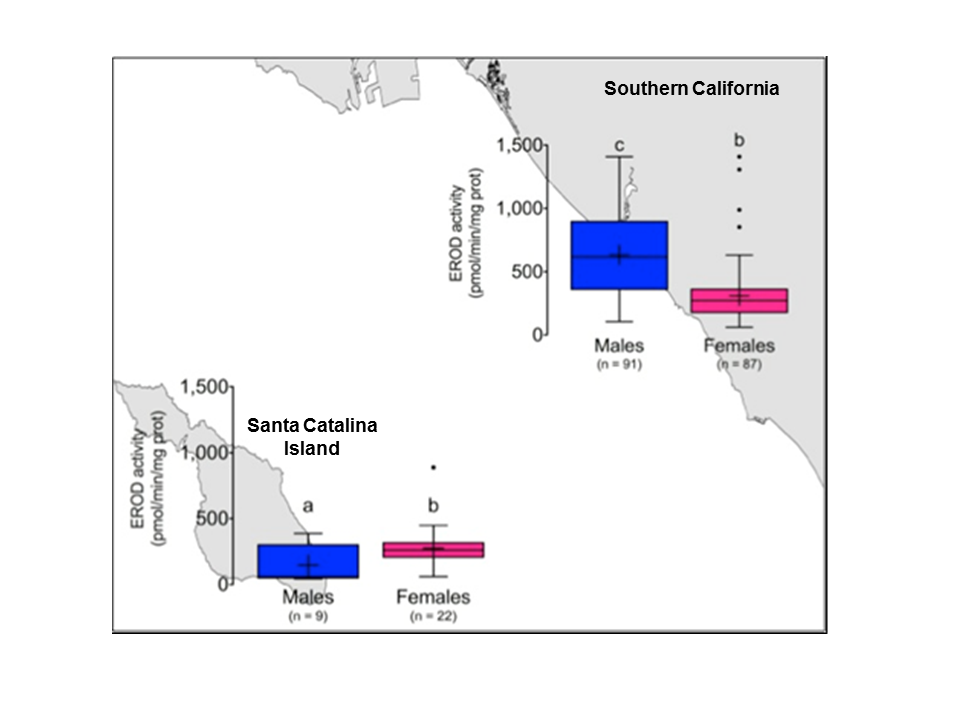
Lyons et al. 2014 found that EROD activity is higher in adult males than females. Adult females have lower concentrations of PCBs when compared to males; however, female EROD activity decreased with disc width despite significant increases in PCB concentrations.
The lack of a difference in EROD activity in mainland and Catalina island females despite differences in contaminant concentration suggests an estrogenic downregulating effect in female round stingrays.
Habitat Use and Exposure
Contaminant profiles varied between adult and juvenile age classes for each sex indicating that contaminant profiles can be used to examine habitat use. Mature females in particular sexually segregate from males after mating and aggregate in calm, estuarine habitats which are subject to a high input of urban runoff following a storm which could explain observed differences in the proportion of contaminant groups tested.
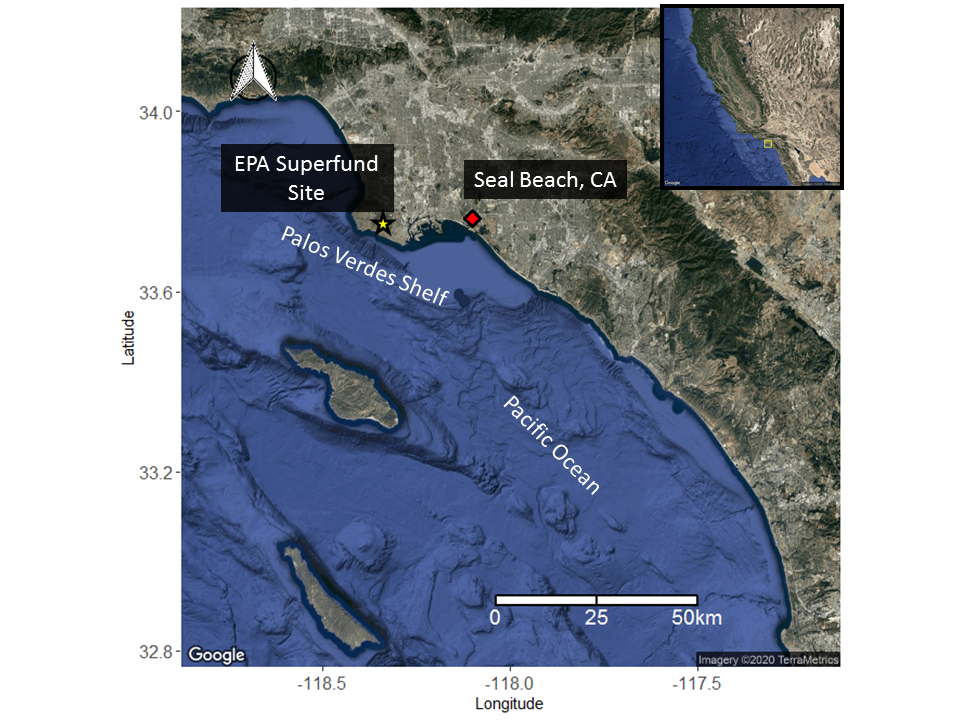
Differences in contaminant concentration were also observed in the Catalina island population of round stingrays as females segregated in an area that had low tidal flushing and was subject to greater human activity. Overall though, environmental exposure is significantly reduced at Santa Catalina Island compared to the mainland.
Mainland populations of round stingrays exhibit low proportions of DDTs indicating that round stingrays do not utilize areas near the Palos Verdes shelf where a large source of DDT is still present in the sediment from human activity.
Immune Response and Exposure
To further determine if chronic contaminant exposure produces significant negative physiological impacts on round stingrays, Sawyna et al. 2016 examined whether round stingrays exhibit impaired immune function due to high contaminant exposure. Results showed that exposed rays experienced higher immune cell proliferation and significantly higher rates of phagocytosis in multiple tissues, both biomarkers of immune impairment. This study provides some of the first evidence of immunostimulation correlated with organochlorine contaminants in rays.